

Next: WaterQuality Activities
Up: Water Quality Measurements
Previous: Water Quality Measurements
Subsections
 Solvent
Solvent
- Water is an excellent solvent, which means
that many chemicals dissolve easily into it
(referred to as dissolved species, Fig. 4.2).
 Dipolar
Dipolar
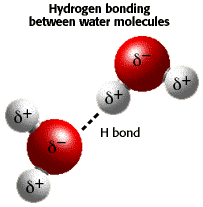
- Water molecules have one end that is more negatively
charged, and one end more positively charged
(Fig. 4.1), allowing it to bond or complex
with most charged species and with itself.
 Benefits
Benefits
- Makes water a useful cleaning agent, and important
transporter of chemicals in all living things
 Costs
Costs
- Water can easily be contaminated with undesirable
chemicals
Figure 4.1:
Dipolar
nature of water molecule and intermolecular hydrogen bond. The two
white hydrogens are more positive than the red oxygen (which is not
sharing two of its electrons).
|
|
It gets more complicated though, because
 Dissociation
Dissociation
- each water molecule can ``come apart'' (termed
dissociation or ionization of water)
 Acid-Base
Acid-Base
- dissociation creates acids (the H
 ion) and
bases (the OH
ion) and
bases (the OH ion)
ion)
 Acid-Base Reactions
Acid-Base Reactions
- the exchange (i.e. donation by the acid
or acceptance by the base) of the proton (H
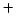 ) is the basis of
many chemical reactions (acid-base reactions),
especially in water
) is the basis of
many chemical reactions (acid-base reactions),
especially in water
 Examples
Examples
- e.g. eating citric acid (tangy sensation), using
muriatic acid on concrete (dissolves stains that water alone can't
get out), taking ``Tums'' (a base) to neutralize gastric acid
 pH
pH
- the measure of concentration of protons (H
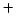 ion) in
water, or essentially the strength of the proton donation
reaction.
ion) in
water, or essentially the strength of the proton donation
reaction.
 pH Definition
pH Definition
- pH is the negative logarithm of the
concentration of H
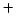 . So an acid has low pH, and therefore high
concentrations of H
. So an acid has low pH, and therefore high
concentrations of H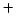 , and can participate more readily in
reactions that require donation of a proton.
, and can participate more readily in
reactions that require donation of a proton.
 neutral pH
neutral pH
- at neutral pH there are equal ``concentrations''
of H
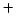 and OH
and OH in the solution. At room temperature neutral
pH is 7. ``Neutral'' really means that there is equal tendency
solution for donation or acceptance of protons.
in the solution. At room temperature neutral
pH is 7. ``Neutral'' really means that there is equal tendency
solution for donation or acceptance of protons.
 Consequences
Consequences
- for example metals (which can often be toxic)
tend to be immobile in acid environments. If we want to
understand the chemical state of a water, we must measure its pH
as well as concentrations of dissolved species.
Figure 4.3:
Dissociation reaction of H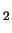 O.
O.
|
|
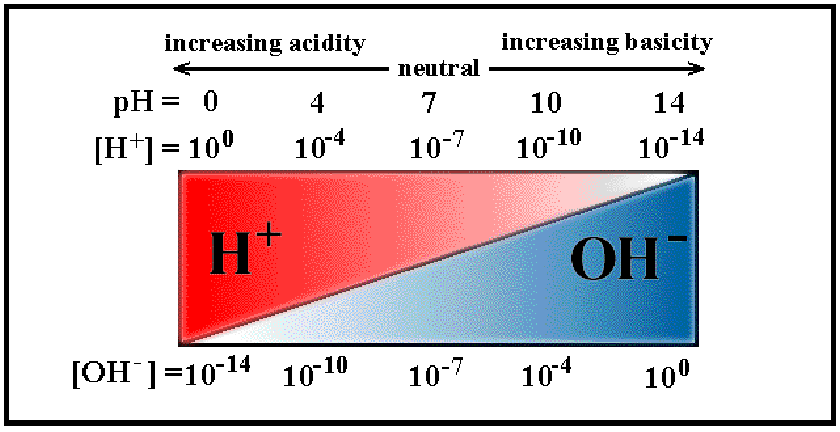
Figure 4.4:
The pH scale, where high pH indicates high
concentrations of protons (H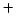 ions), and a high potential for
proton donation.
ions), and a high potential for
proton donation.
|
|
We must also measure temperature of our water because:
- Equilibrium state in all chemical reactions depends on
temperature
- e.g. its easier to dissolve sugar in a hot drink than a cold
one, because the solubility of sugar (and most chemicals) in
water increases with temperature
- similarly, pH depends strongly on temperature Fig. 4.5
Figure 4.5:
Variation of pH vs. temperature.
|
|
Since many dissolved species of interest are metals, we must also
characterize the oxidation (``rusting'' and transport) potential of
the water
- rust is the familiar process of iron (Fe
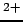 )
oxidizing (donating electrons to oxygen) to form
(Fe
)
oxidizing (donating electrons to oxygen) to form
(Fe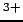 )
)
- in general the reduced form of metals (e.g. Fe
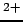 ) is more
mobile/soluble in water
) is more
mobile/soluble in water
- oxidation state is best characterized by oxidation
potential or Eh
- we'll use a proxy for this, which is to measure the
dissolved oxygen or DO of the water
- waters with high DO are good for animals (lots of O
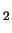 to
breathe), and don't transport as many metals
to
breathe), and don't transport as many metals
- waters with lots of dead organic matter consume oxygen by
converting the carbon to CO
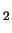 , and therefore tend to have low
values of DO
, and therefore tend to have low
values of DO
- DO saturation is elevation and temperature dependent
Other Parameters
Some other parameters are useful:
 Turbidity
Turbidity
- the cloudiness of the water. An indicator of
suspended particulates, which can transport bacteria. EPA
Drinking Water limit is 1 NTU (nephelometric turbidity units).
Easily controlled by settling or filtration.
 Electrical Conductivity
Electrical Conductivity
- is an indication of the amount of
dissolved ions. The more ions, the easier it is for electricity
to move through the water (essentially electrons hop from ion to
ion, the more ions, the easier that is). Typical values for
drinking water are around 300
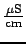 (e.g. Fine Waters website).
Specific Conductance is the electrical conductivity
adjusted to 25
(e.g. Fine Waters website).
Specific Conductance is the electrical conductivity
adjusted to 25
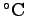 , to allow direct comparison of waters that
have differing temperatures.
, to allow direct comparison of waters that
have differing temperatures.
 Salinity
Salinity
- also known as TDS or total dissolved
solids, given in ppm. Often inferred from specific conductance or
computed as the sum of all dissolved species. The Safe Drinking
Water Act limit for TDS is 1000 ppm.
 Nitrate
Nitrate
- usually a remnant of fertilizer application, maximum
allowed in drinking water is 10ppm. Measured using
spectrophotometer (colorimeter)
 Hardness
Hardness
- the potential to form carbonate scale, this is the
sum of Ca and Mg, usually dominated by Ca
 Alkalinity
Alkalinity
- essentially the concentration of the anion
HCO
 , which is dominant in many surface water systems.
Alkalinity is reported as ppm CaCO
, which is dominant in many surface water systems.
Alkalinity is reported as ppm CaCO



Next: WaterQuality Activities
Up: Water Quality Measurements
Previous: Water Quality Measurements
GEOS 3110 Professor's Notes, Summer 2007
Dr. T. Brikowski, U. Texas-Dallas. All rights reserved.